CMSC
-0.0100


Japan has approved ground-breaking stem-cell treatments for Parkinson's and severe heart failure, one of the manufacturers and media reports said Friday, with the therapies expected to reach patients within months.
Pharmaceutical company Sumitomo Pharma said it received the green light for the manufacture and sale of Amchepry, its Parkinson's disease treatment that transplants stem cells into a patient's brain.
Japan's health ministry also gave the go-ahead to ReHeart, heart muscle sheets developed by medical startup Cuorips that can help form new blood vessels and restore heart function, media reports said.
The treatments could be on the market and rolled out to patients as early as this summer, reports said, citing the health ministry, becoming the world's first commercially available medical products using (iPS) cells.
Japanese scientist Shinya Yamanaka won the Nobel Prize in 2012 for his research into iPS, which have the potential to develop into any cell in the body.
"I hope this will bring relief to patients not only in Japan but around the world," health minister Kenichiro Ueno told a press conference.
"We will promptly carry out all necessary procedures to ensure it reaches all patients without fail."
In a statement, Sumitomo Pharma said it had obtained "conditional and time-limited approval" for the manufacture and marketing of Amchepry under a system which is reportedly designed to get these products to patients as quickly as possible.
The approval is a kind of "provisional license", the Asahi newspaper said, after the safety and efficacy of the treatment was judged based on data from fewer patients than in ordinary clinical trials for drugs.
A trial led by Kyoto University researchers indicated that the company's treatment was safe and successful in improving symptoms.
The study involved seven Parkinson's patients aged between 50 and 69, with each receiving a total of either five million or 10 million cells implanted on both sides of the brain.
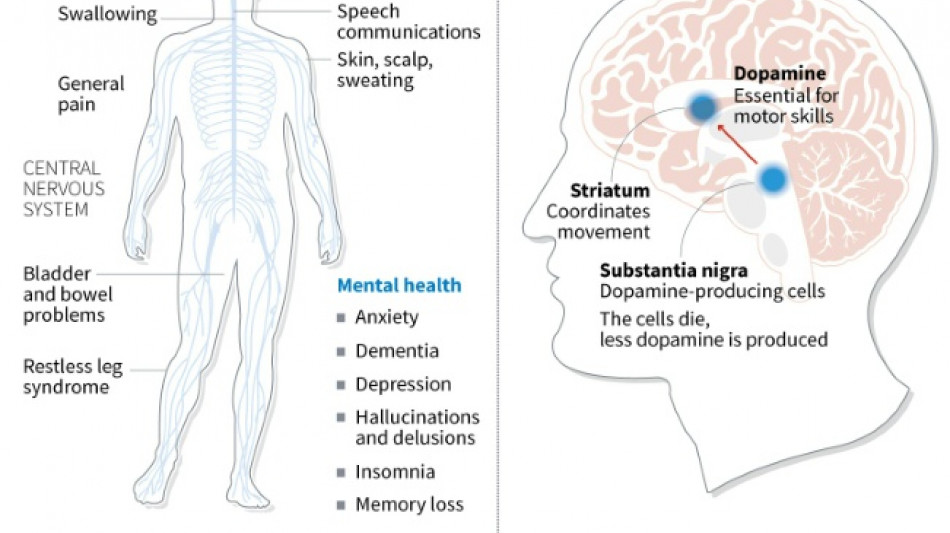
The iPS cells from healthy donors were developed into the precursors of dopamine-producing brain cells, which are no longer present in people with Parkinson's disease.
The patients were monitored for two years and no major adverse effects were found, the study said. Four patients showed improvements in symptoms.
Parkinson's disease is a chronic, degenerative neurological disorder that affects the body's motor system, often causing shaking and other difficulties in movement.
Worldwide, about 10 million people have the illness, according to the Parkinson's Foundation.
Currently available therapies "improve symptoms without slowing or halting the disease progression," the foundation says.
iPS cells are created by stimulating mature, already specialised, cells back into a juvenile state -- basically cloning without the need for an embryo.
The cells can be transformed into a range of different types of cells, and their use is a key sector of medical research.
K.Ibarra--TFWP